 I have followed all appropriate research reporting guidelines and uploaded the relevant EQUATOR Network research reporting checklist(s) and other pertinent material as supplementary files, if applicable. I confirm that any such study reported in the manuscript has been registered and the trial registration ID is provided (note: if posting a prospective study registered retrospectively, please provide a statement in the trial ID field explaining why the study was not registered in advance). S816-830-reactive T cells initially contributed up to 100 of the S-IIreactive CD4+ T cells but their proportion decreased during the course of the S-II. 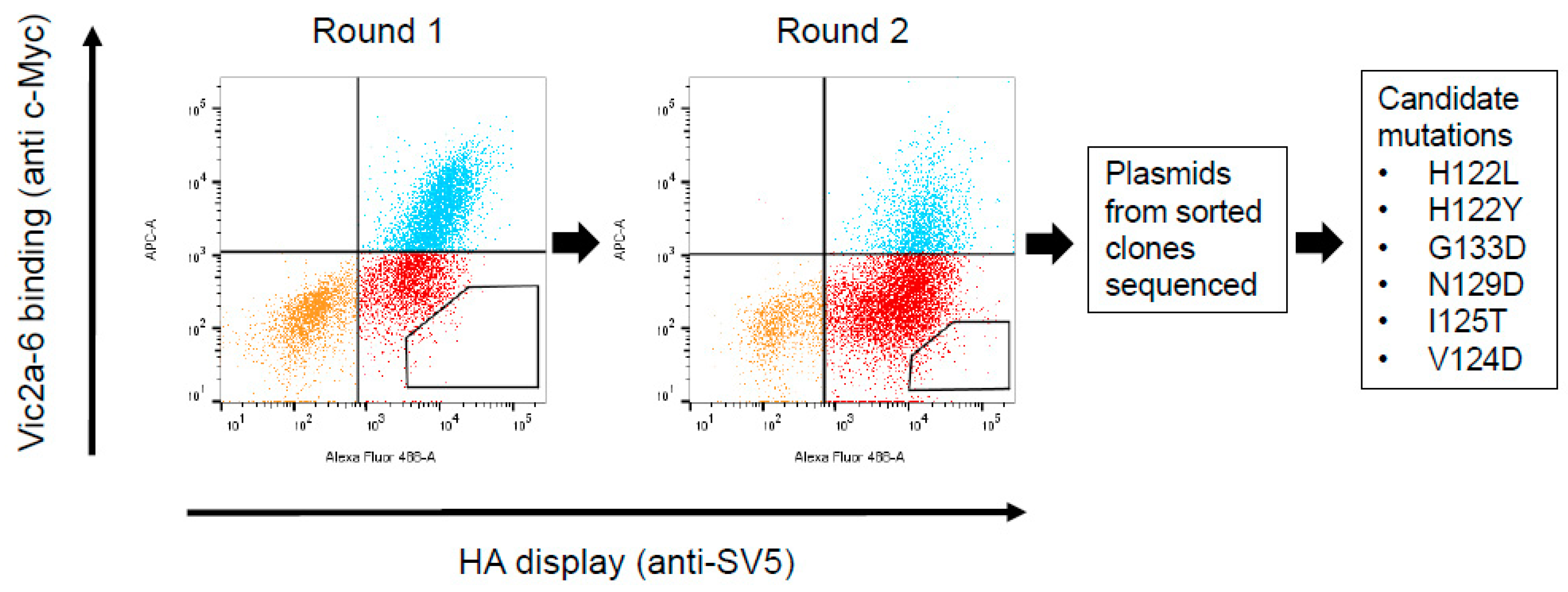
This same crossreactivity can also be a significant problem with immunoassays if a highly specific measure of only a single analyte is needed. I understand that all clinical trials and any other prospective interventional studies must be registered with an ICMJE-approved registry, such as . This crossreactivity is utilized in drug screening methods that provide rapid turnaroundtime but less overall certainty to the specific identity of the measured analyte. I confirm that all necessary patient/participant consent has been obtained and the appropriate institutional forms have been archived, and that any patient/participant/sample identifiers included were not known to anyone (e.g., hospital staff, patients or participants themselves) outside the research group so cannot be used to identify individuals. The Institutional Review Board of Emory University gave ethical approval for this work. between degree of immunological cross-reactivity and degree of amino acid sequence similarity, with the limitation that proteins differing from each other by 40 or more in sequence exhibited no cross-reactivity in micro-complement fixation tests. The details of the IRB/oversight body that provided approval or exemption for the research described are given below: I confirm all relevant ethical guidelines have been followed, and any necessary IRB and/or ethics committee approvals have been obtained. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. This study was funded by the National Institute of Allergy and Infectious Diseases. 
The authors have declared no competing interest. We demonstrate that neuroinvasive West Nile virus infection leads to the development of non-neutralizing, cross-reactive IgG antibodies to dengue and Zika viruses capable of causing antibody dependent enhancement in vitro of dengue virus and leads to the formation of flavivirus cross-reactive memory B cells in some patients. Antibody dependent enhancement has been well described between Zika and dengue viruses, but is poorly characterized between West Nile and dengue viruses. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
